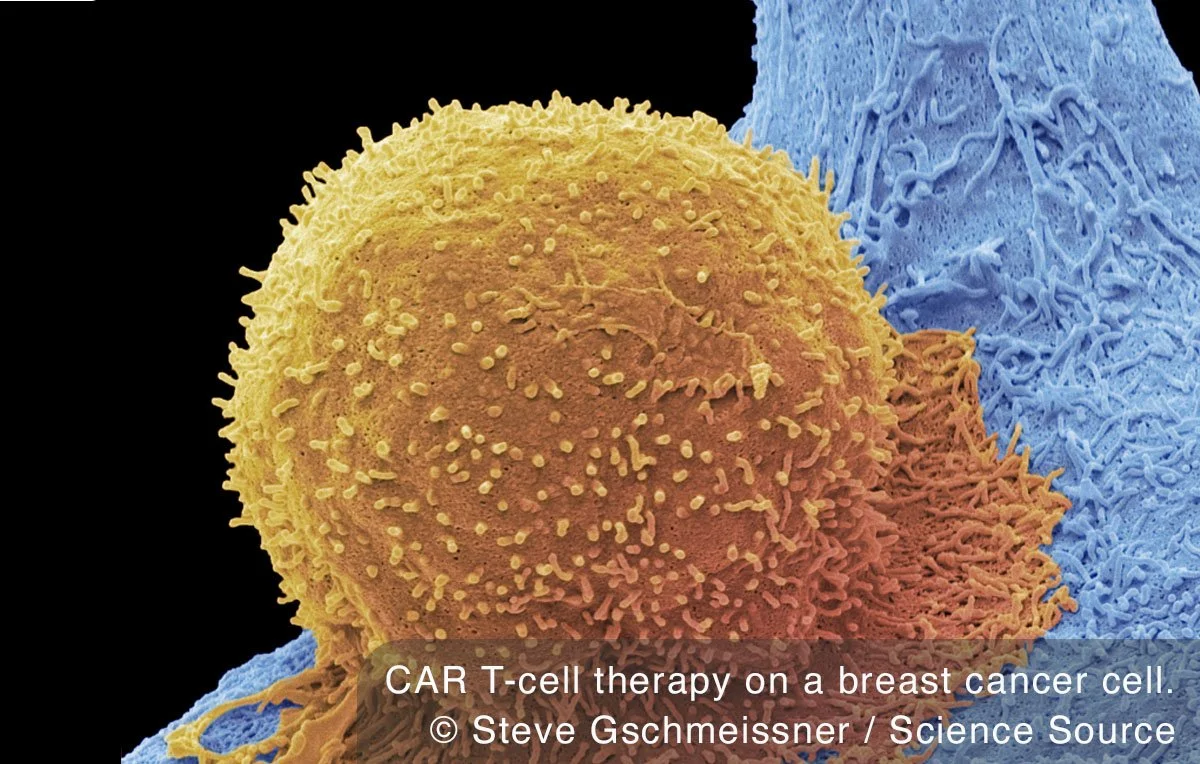
What if your own immune cells could be trained to recognize and destroy cancer?
This is no longer a futuristic idea—it’s happening right now.
In 2017, the U.S. Food and Drug Administration approved CAR T-cell therapy, a groundbreaking treatment often described as a “living drug.” Since then, more than 35,000 patients have received it, offering new hope where traditional therapies have failed.
GALLERY OF CAR T-CELL THERAPY IMAGES
How It Works
CAR T-cell therapy harnesses the power of your immune system in a highly personalized way:
First, doctors collect a blood sample and isolate T-cells—white blood cells that play a central role in immune defense. In the lab, these cells are genetically modified to produce Chimeric Antigen Receptors (CARs), which allow them to recognize specific cancer cells.
Once reprogrammed, the cells are multiplied and infused back into the patient’s body. From there, they actively seek out and destroy cancer cells with precision.
A Targeted Approach
Unlike chemotherapy, which can damage healthy cells along with cancerous ones, CAR T-cell therapy is designed to target only cancer cells. This precision reduces collateral damage and can lead to more effective outcomes.
The therapy has shown particular success in treating blood cancers such as leukemia and lymphoma—especially in patients who have not responded to standard treatments.
Long-Term Impact
One of the most exciting aspects of CAR T-cell therapy is its potential durability. Many patients experience remission lasting three to four years, with some cases extending up to nine years and beyond.
With advances in CRISPR, these therapies are becoming increasingly precise and effective. Researchers are now expanding their use beyond blood cancers to target solid tumors such as breast, prostate, and sarcomas, with ongoing development for challenging conditions including glioblastoma, melanoma, liver cancers, and even certain autoimmune diseases.
A New Era in Cancer Treatment
CAR T-cell therapy represents a fundamental shift in how we approach cancer—from broadly attacking the disease to precisely engineering the body’s own defenses.
What was once rapidly fatal can now, in some cases, be managed for years—or even decades.
Explore the Science Visually
View compelling micrographs of CAR T-cells attacking cancer, alongside detailed illustrations and diagrams that bring this process to life at Science Source. Our collection of medical imagery is supported by accurate, robust metadata—helping educators, researchers, and communicators tell these powerful stories with clarity and credibility.
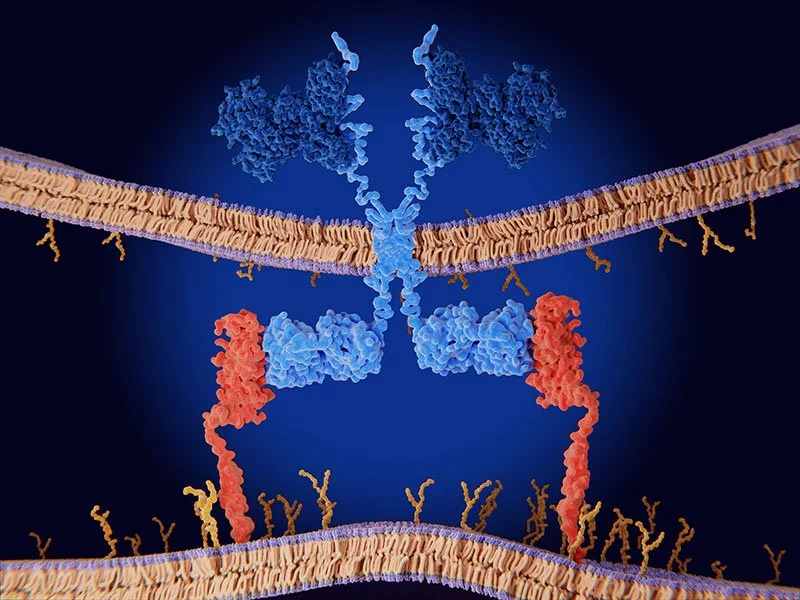
Chimeric antigen receptor (CAR) therapy.
Engineered receptor (light blue) on the surface of a T-lymphocyte bind specifically to CD19-antigen molecules (red) on a leukemia cell. © Juan Gaertner / Science Source
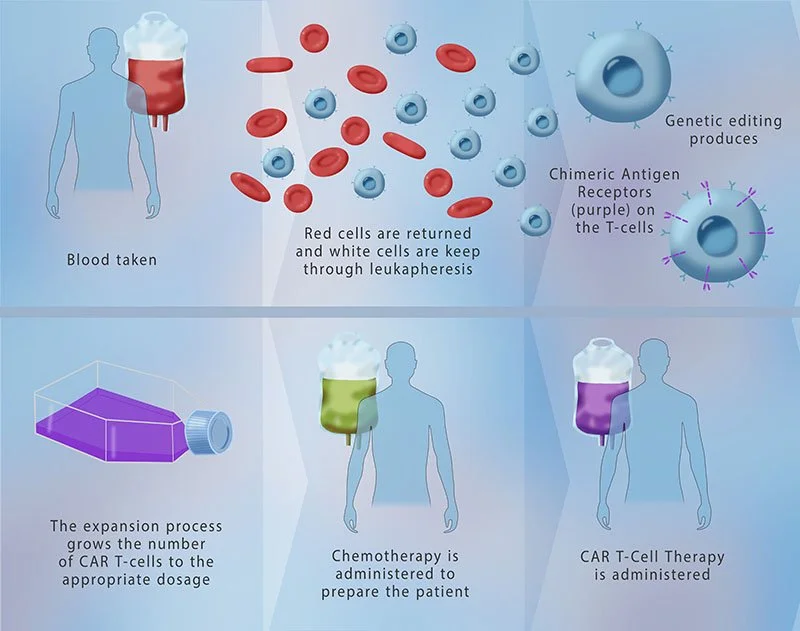
Diagram of the (CAR) T-cell Therapy Process
A diagrammatic illustration of chimeric antigen receptor (CAR) T-cell therapy. The process begins with the extraction of blood from the patient. Through leukapheresis, T-cells are isolated. © Monica Schroeder / Science Source
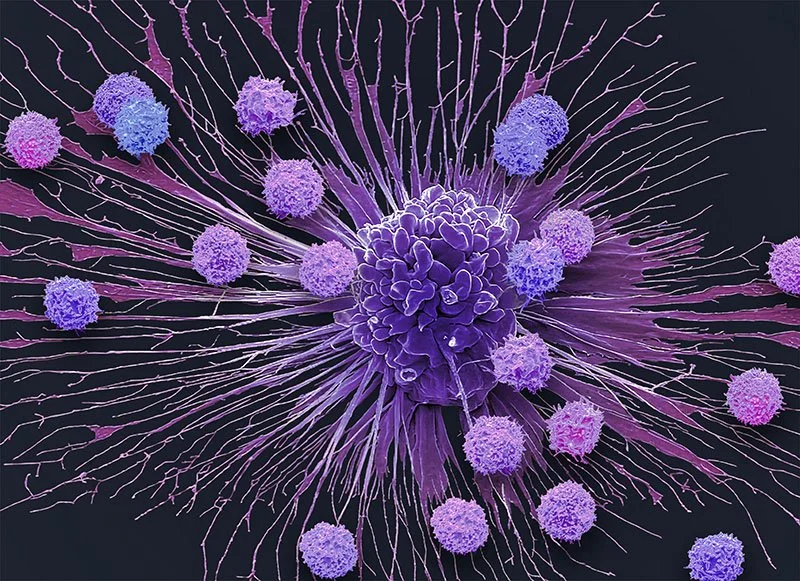
T-cells Surrounding an Apoptotic Hela Cervical Cancer Cell.
Scanning electron micrograph (SEM) of T-cells (small round) and an apoptotic cervical cancer cell (HeLa). T-cells are a component of the body's immune system. © Steve Gschmeissner / Science Source